4 minute read
Understanding the Bone Marrow Niche: A Key to Advancing Hematological Research
May 9, 2025
March 13, 2025
February 17, 2025
March 13, 2025
February 17, 2025
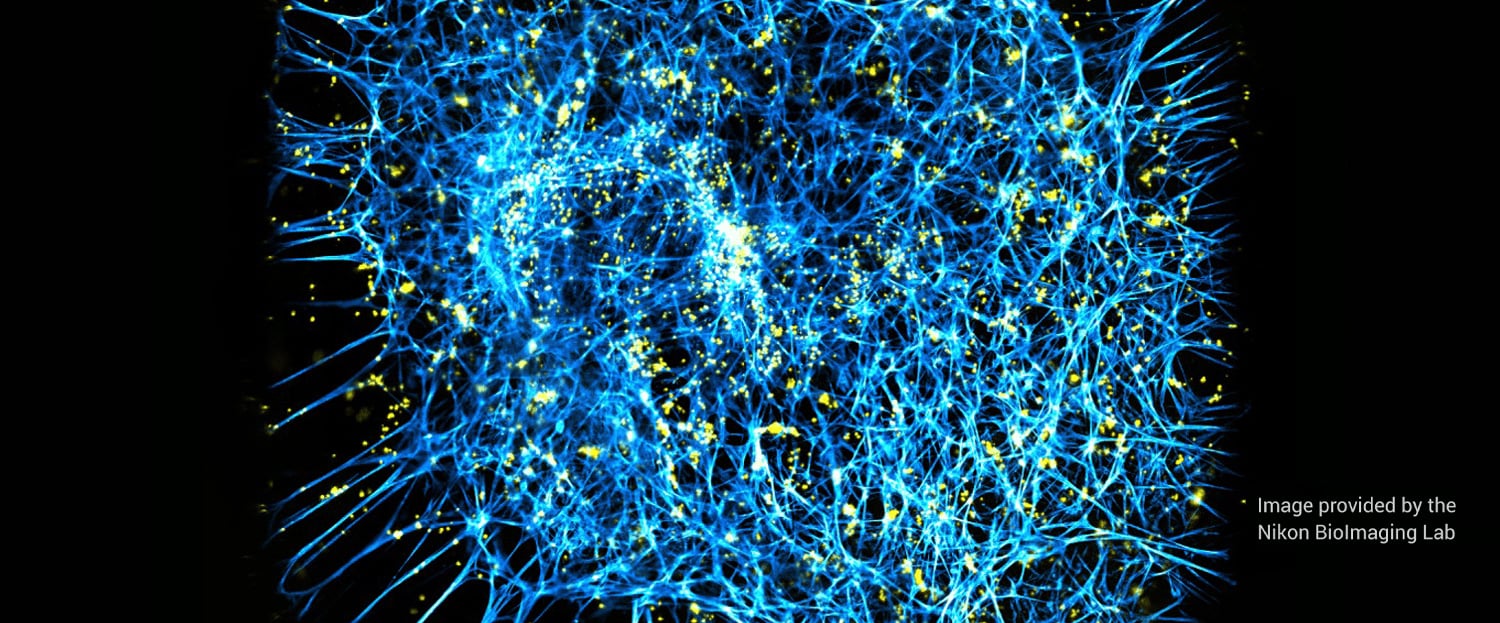
4 minute read
May 9, 2025






1 minute read
March 27, 2025
© 2025 Crown Bioscience. All Rights Reserved.
Privacy Policy | Imprint | Terms of Service | Privacy Preferences


© 2025 Crown Bioscience. All Rights Reserved. Privacy Policy
2025-03-03
2022-12-16