I’m a firm believer in the use of animal models in medical research. This is so for many reasons. A chemist and mentor of mine once said “to truly do drug discovery you absolutely need a liver and circulatory system”.
I’m a firm believer in the use of animal models in medical research. This is so for many reasons. A chemist and mentor of mine once said “to truly do drug discovery you absolutely need a liver and circulatory system”.
For example, in oncology, a cancer isn’t merely a collection of cancer cells. It is a complex, albeit disorganized tissue, with many interacting cell types. I myself have witnessed the critical importance of the interactions between cancer cells and their surroundings, including endothelial cells, immune cells, and other supportive stromal cells. The activity of one drug program I worked on absolutely depended upon these complex interactions.
So, when I look to the burgeoning field of “organs-on-a-chip”, please humor me, I’m a bit skeptical. The development and functioning of an organ, any organ, is a truly astonishing feat of biology – biology with a capital “B”. To me, this process is so astounding that I challenge the hubris in that we think we can recapitulate even a fraction of this, ex vivo and on a fabricated chip.
And yet, as I explore this field, I find myself amazed.
Organ-on-a- Chip Technology Captures the Mechanical Features that Cells Experience
At the interface of materials, microfluidics, and cell biology, organ-on-a-chip technology aims to recapitulate many of the features of the organs of the body that cannot be captured in standard two-dimensional, monolayer tissue culture. It may be surprising to learn, but the chief feature that organ-on-a-chip technology captures is the mechanical features that cells in organs experience.
Flow and shear stress experienced by endothelial cells, expansion and contraction of airways experienced by lung epithelium, and wave like peristaltic motion experienced by intestinal epithelium are all sources of fundamental signals that define and shape the cells of these organs. Under these conditions in a chip format, for instance, lung epithelia produce mucus and develop cilia, which beat in a uniform direction.
I encourage all of you reading this to watch the following video of a lecture given by Donald Ingber, a pioneer not only in the area of organ-on-a-chip technology, but someone who has spent the better part of their career understanding the mechanical features of biology.
Multitude of Applications to Drug Development including Immuno-Oncology
It isn’t too hard to imagine the multitude of applications of an ex vivo technology that better recapitulates cell biology at the organ level. Delivering drugs to the CNS is made challenging by the blood-brain-barrier. Better models of this structure will lead to better CNS drugs.
The ability to peer into the spatial and temporal interplay of cancer cells and immune cells, with clear resolution, will potentially help us understand phenomena such as T cell exhaustion. This in turn will potentially lead to a better understanding of therapeutic response heterogeneity in the immuno-oncology clinic.
New Technology to Minimize Animal Model Use
One further benefit to the continued development of this technology is the minimization of the use of animals in medical research. While I believe that “you need a liver and circulatory system to do drug discovery” that doesn’t necessarily mean the liver and circulatory system actually have to be attached to an animal.
Future Prospects for Improving Drug Development
If you watched the video, you will have noted that Dr. Ingber strongly feels that the current drug development model is “broken”. The fact is drug development is difficult. While it is true that sometimes models do not predict clinical outcomes, sometimes they do.
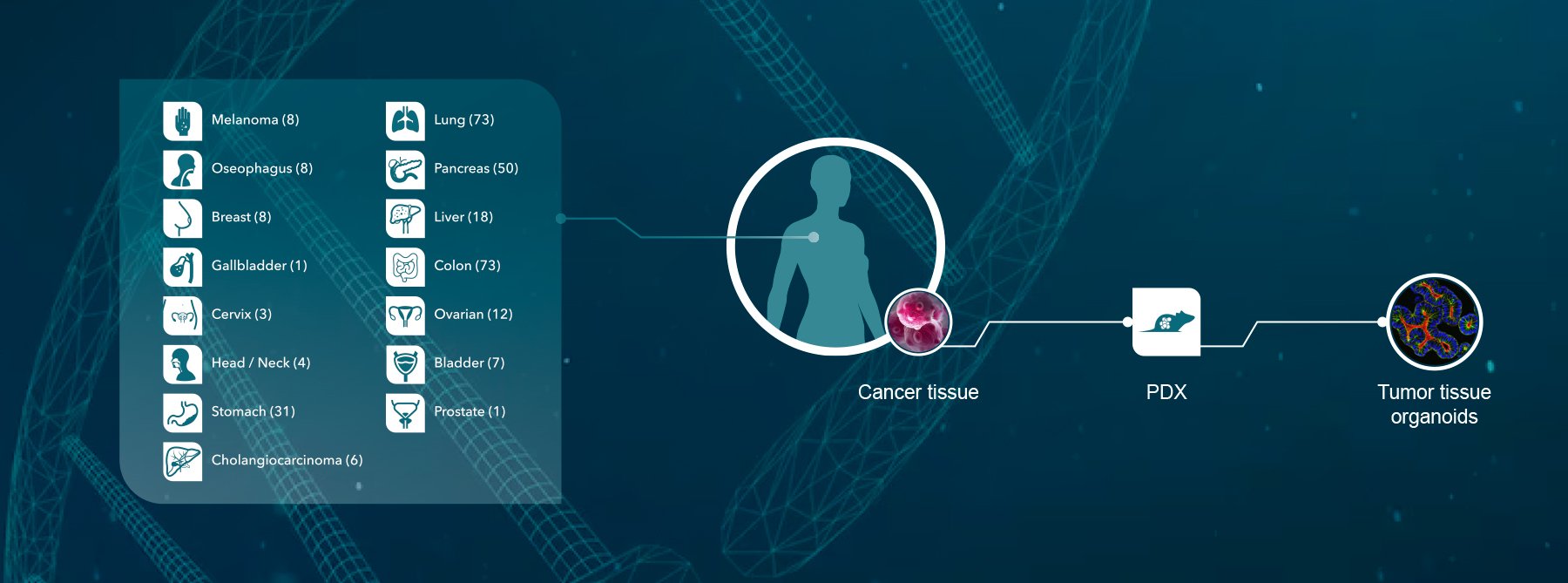
With continued advances in models of disease – for instance, the use of PDX in vivo oncology models or the continued exploration of organ-on-a-chip technology – drug developers will be better armed with the tools to make more informed decisions leading to improved translational fidelity.