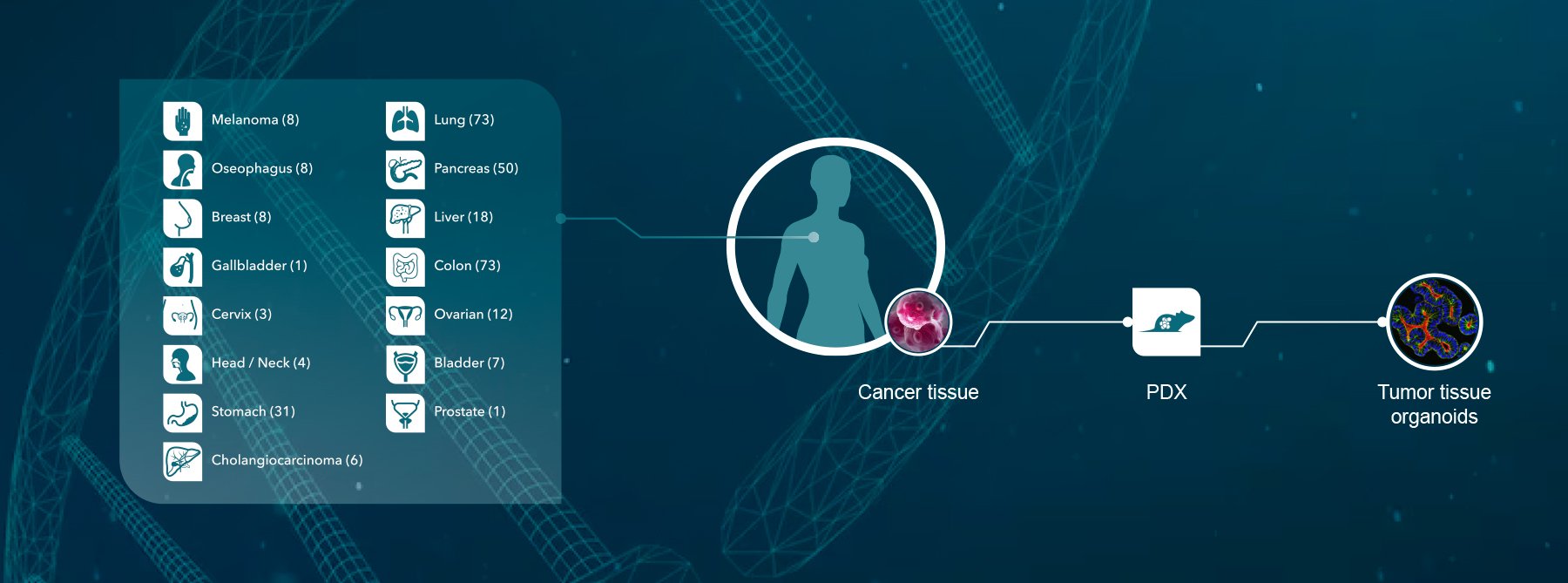
One of the main goals of translational research for oncology is to discover biomarkers which predict clinical efficacy. With the world’s largest collection of over 1,000 well-characterized PDX models, HuPrime®, and HuBase™ (a comprehensive database of genotypic, pharmacological, and patient clinical data on HuPrime models), Crown Bioscience is enabling drug discovery organizations to perform human surrogate Phase II-like mouse clinical trials, also known as HuTrials™, to identify suitable disease indications and discover and validate predictive biomarkers for their therapies. At the recent AACR annual meeting, Crown Bioscience presented an original research poster on the parameters influencing the design of a randomized, controlled, and statistically powered HuTrial.
What can a HuTrial do for you in drug discovery research?
The most significant cost driver for drug development is the high failure rate in late stage clinical development. The need to reduce this drug attrition is especially acute in the field of oncology where drugs often fail because of a lack of efficacy rather than toxicity. The successful development of drugs like trastuzumab, imatinib, and gefitinib has demonstrated the critical need to identify biomarkers in order to select patients which are most likely to benefit from a given drug treatment. Using PDX models in a Preclinical Phase II-like trial or HuTrial, you can evaluate efficacy in models that come from the direct lineage of a patient’s tumor, select patient responders and non-responders, identify genetic signatures of disease, and discover and validate predictive biomarkers for your agent before a single patient is dosed. HuTrials can then be used to position, or re-position, your drug in the marketplace.
How many models should you run in a HuTrial?
We advise to run as many models as possible, to represent the wide range of diversity that is seen within a patient population. Running a large number of models will also yield better powered statistical results. The minimum number of models needed is influenced by many factors including the question you want to answer in your trial, how many drugs you are testing, and how potent your drug is. For example, in a single agent HuTrial, the potency of the treatment affects how many models need to be used based on statistical power. On average, we have run 30 to 100 models in previously completed HuTrials.
And how many mice should be used per arm?
Again, this question does not have a simple answer, although on average 5 mice per arm have often been used in previous HuTrials. The number of mice needed per arm varies depending on the number of models used and the potency of the drug to be tested. Increasing the number of mice per arm decreases variations in tumor volume ratio (e.g. TGI) for each model. Increasing the potency of the drug also decreases this variation, whilst increasing the number of mice per arm will increase the statistical power of the study results. Crown Bioscience work with each client to design their HuTrial to suit their individual needs and can fully advise on all of the technical detail surrounding numbers of models and mice to use dependent on your study design and drug to be trialled.
Can HuTrial endpoints be translated to the clinical setting?
Crown Bioscience design your HuTrial to match your clinical trial needs as closely as possible, providing objective response or “survival” endpoints as required. For objective response, the RECIST criteria (complete response or CR, partial response or PR, stable disease or SD, and progressive disease or PD) can be calculated and used to mimic the clinic endpoint. Data can also be presented in waterfall plots as seen in human studies. Endpoints, including overall survival or “time to event” (such as progression free survival), can be analyzed and presented with Kaplan-Meier plots for HuTrials. Crown Bioscience have fully validated HuTrials to ensure that these endpoints are related to the drugs tested rather than the tumor growth rate. Efficacy within a HuTrial has been shown not to correlate with tumor growth, meaning it is a true property of a drug and not a tumor growth readout.
Why should I run my HuTrial with Crown?
HuTrials are possible because of Crown Bioscience’s ability to leverage the world’s largest commercial collection of genomically characterized PDX models, which is large enough to be truly reflective of the patient population. Appropriate models that meet a client’s criteria of responder and non-responder can be easily selected using HuBase. However, just having the largest collection of models is not enough. At Crown Bioscience many of our models are in constant passage and we have the operational capacity to screen multiple models simultaneously and perform large numbers of HuTrials every year. This means that we can perform your HuTrial in months instead of years, in a cost-effective and streamlined way.
Contact us at busdev@crownbio.com to talk to our experts about how HuTrials can drive forward your drug development research.