Immunotherapy is at the forefront of oncology research, with many companies focusing on the same specific targets in multiple cancer types. The race has been on for the first US approval of an anti-PD-1 agent, and Merck have proved victorious with the FDA saying yes to Keytruda (pembrolizumab) in advanced melanoma just last week.
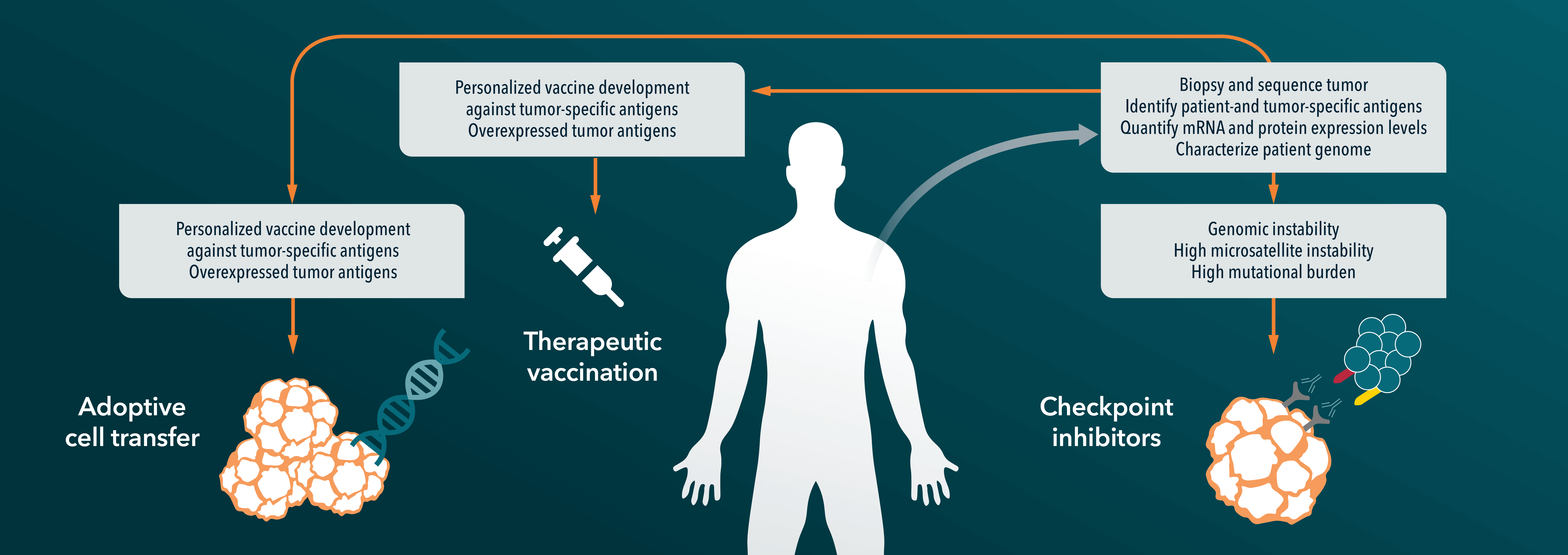
Anti PD-1/PD-L1 agents have been a hot topic in immunotherapy in oncology this year. Programmed death receptor 1 (PD-1) is an inhibitory receptor expressed on the surface of human immune cells. Ligands for the receptor, PD-L1 and PD-L2, are found on cells throughout the body, and when the ligand and receptor interact normally they block the function of T cells. However, tumor cells can also express PD-L1, which can then interact with the PD-1 receptor and reduce the natural immune response to cancer. Both anti-PD-1 and PD-L1 agents block this interaction, boosting a patient’s own T cell function to fight back against the invading tumor.
Crown Bioscience featured clinical results on anti-PD-1/PD-L1 agents as our highlight of the ASCO conference earlier this year, reporting that novel agents were being evaluated in over 30 different cancer types. Merck and Bristol-Myers Squibb are two of the companies at the cutting edge of research, and Merck were given an FDA priority review for pembrolizumab in metastatic melanoma following previous treatment with ipilimumab (another immunotherapy which blocks the “immune checkpoint” molecule CTLA-4), with a target review date of 28th October. The FDA have reached their decision a lot more quickly than that though, and last week approved the newly renamed Keytruda for patients with advanced melanoma who no longer respond to other treatments. The Bristol-Myers Squibb agent nivolumab was approved in Japan in July, but the FDA decision marks the first approved anti-PD-1/PD-L1 agent in the US or Europe. Nivolumab is also expected to be reviewed by US regulators in the coming months.
This is excellent news for melanoma patients in the US. Melanoma is the most malignant form of skin cancer, and while it accounts for less than 2% of all US skin cancer cases it causes the vast majority of skin cancer deaths. Due to increased sunbed use and holidays in the sun (and also better detection), melanoma incidence rates have been steadily increasing in the US over the last 30 years. It is estimated that this year alone over 76,000 new cases of melanoma in the US will be diagnosed, with 9,700 deaths from the disease. Survival rates for advanced cases used to be under 1 year, but in the Keytruda clinical trial data presented at ASCO 69% of patients receiving the treatment survived 1 year, with 62% still alive at 18 months. Only 4% of subjects in the trial discontinued due to treatment-related adverse events.
Merck are now going on to look at whether the drug could be effective in early stage patients, and are still testing in a range of cancer types, as well as trialing a variety of combination therapies. Many other companies are still working on PD-1 pathway drugs, with some analysts reporting that this class of agents will become blockbusters generating more than $30 billion in annual sales worldwide by 2025.
It should be noted that Keytruda and other immunotherapies don’t work in every patient, and more work is needed to find biomarkers which can show which patients are most likely to respond. A second issue with Keytruda and immunotherapeutics in general is price – Keytruda will cost around $12,500 per month, or $150,000 for a one year treatment, with nivolumab costing the equivalent of $143,000 in Japan. These prices are likely to reignite debates on the cost of oncology treatments, and may cause problems in countries which base their approvals solely on cost-benefit analysis.
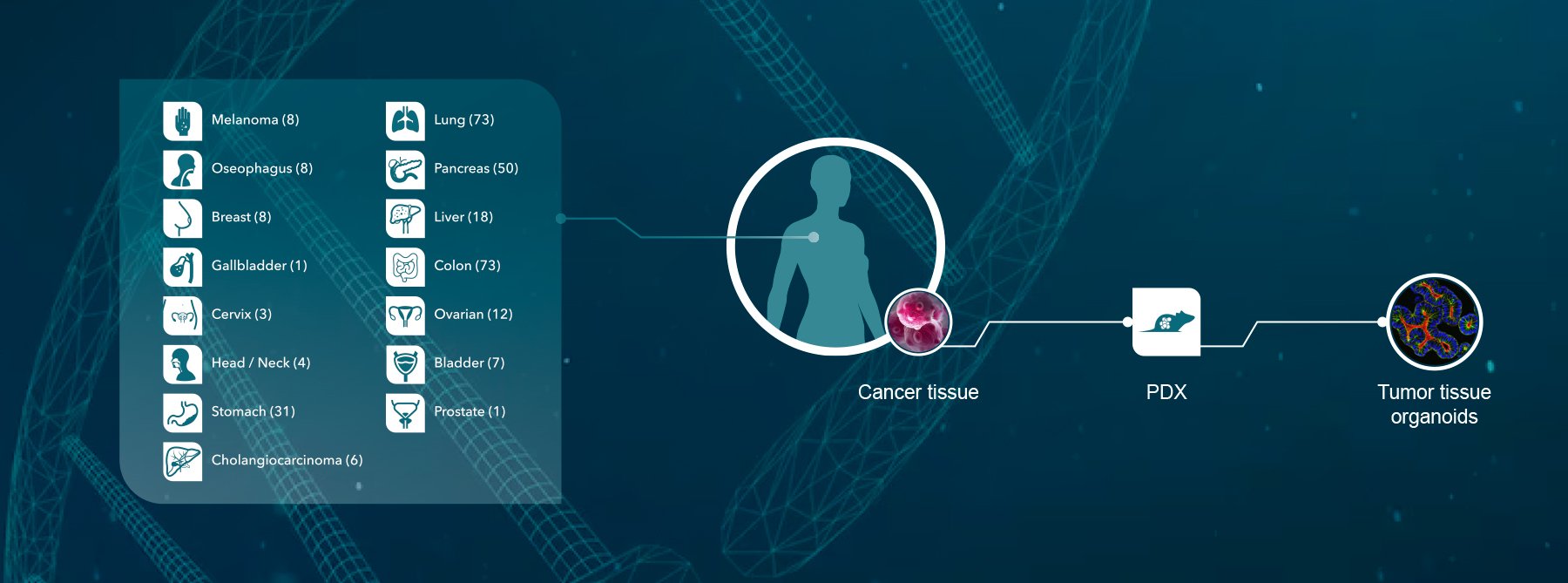
Overall, Crown Bioscience are delighted to see the first US approval for anti-PD-1 therapies. Crown Bioscience support research in immune therapies with a range of immunotherapy platforms (with either murine or human immunity) in a range of different cancer types for use in your research, including syngenic models, GEMM, MuPrime™ (the murine version of HuPrime® which is the world’s largest collection of well-characterized and validated Patient-Derived Xenograft models), HuMice™ (humanized mice produced through inoculating human hematopoietic cells into immunocompromised mice), and MiXeno™ (creating transient human immunity by mixing human peripheral blood mononucleated cells with xenograft models). We also support melanoma and metastatic melanoma research through the use of our clinically relevant Xenograft and PDX models available for drug discovery and translational sciences.
Contact us today at busdev@crownbio.com to discover how we can transform your melanoma and immune therapy research.