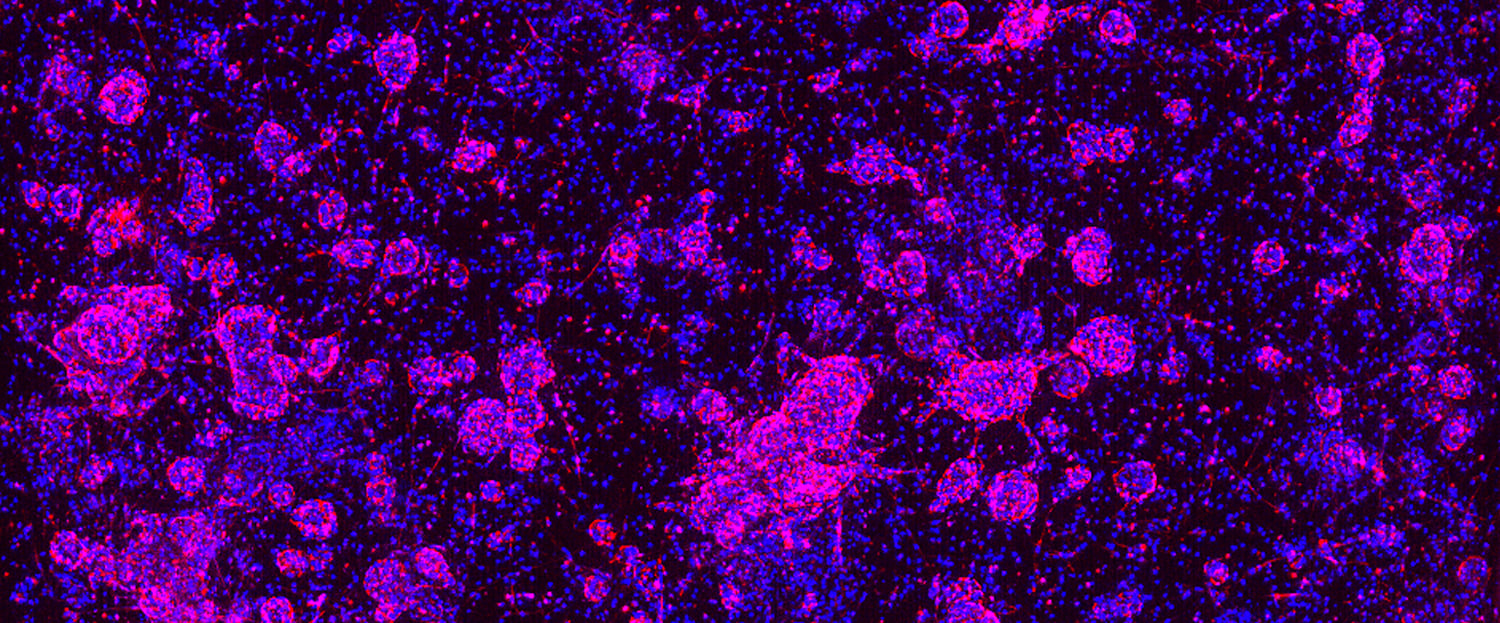
Selecting the right preclinical model to evaluate a drug candidate’s mechanism of action and efficacy can be challenging given the wide variety of available options and their associated time and cost investments. However, selecting the appropriate model can mean the difference between success and failure for a given drug candidate. 3D in vitro organoids are powerful predictive preclinical models that are ideal for bridging the gap between traditional 2D in vitro cell lines and in vivo studies because they offer a more clinically relevant platform than 2D cell lines prior to transitioning to more costly and time-consuming in vivo validation studies.
Published data have confirmed the “biological equivalency” (>90% correlation) in drug response profiles between matched patient-derived xenograft (PDX) models (i.e., the best preclinical in vivo model for predicting clinical efficacy) and 3D in vitro tumor organoids derived from the same tumor. This means that tumor organoids can serve as patient surrogates in high throughput drug screens and other in vitro applications, which can reduce costs and time associated with using in vivo models, while maintaining clinical relevance. This post highlights the value of using highly clinically relevant patient-relevant tumor organoids for advancing your patient-centric research.
What are Tumor Organoids?
Organoids are 3D in vitro models that can be generated from both healthy and diseased tissues. Scientists at Hubrecht Organoid Technology (HUB) have developed specialized protocols to grow organoids directly from a wide variety of primary human tumors (i.e., tumor organoids) that preserve the cancer stem cell (CSC) compartment. These patient-relevant 3D in vitro cultures faithfully recapitulate the phenotypic, morphologic, and genetic features of their original patient tumor over the long term. Data from peer-reviewed studies (for example, here and here) have established HUB tumor organoids as the only clinically relevant 3D in vitro models with the following features:
- Genomic and phenotypic stability in long-term culture and after cryopreservation,
- High clinical predictivity, and
- Ease of scalability relative to in vivo models.
These features mean that tumor organoids can be passaged over long periods of time, biobanked, and later resuscitated from frozen for follow up studies while retaining the phenotypic and genetic features of the parental tumor over multiple generations.
Tumor organoids are produced using tissue directly from patient tumors (i.e., patient-derived organoids [PDOs]) or patient tumor tissue expanded in mice as PDXs (PDX-derived organoids [PDXOs]), and even patient-derived orthotopic xenografts (i.e., PDOXs). Both PDXOs and PDOXs enable the expansion of PDO collections for population studies by generating new tumor organoid models from existing PDXs.
In 2019, Crown Bioscience entered into a strategic partnership with HUB to become the exclusive provider of HUB technology for preclinical cancer drug development and validation.
Should I Use a PDX or Organoid Model?
PDXs are the optimal preclinical in vivo model for predicting clinical efficacy due to their ability to accurately recapitulate the genomic and phenotypic complexity of human tumors. Because PDXs are derived directly from patient tumor tissue, they closely mirror the genetic integrity relative to the original patient tumor. Additionally, collections of PDX models derived from a specific patient population capture the tumor heterogeneity within that population. However, they can be costly, and time consuming to develop and maintain, making them better suited to later stage studies rather than early-stage large scale screens of candidate compounds.
3D in vitro organoids offer the flexibility of in vitro systems, with the predictive power of patient-derived models. Thus, their application extends to a range of in vitro studies, including:
- High-throughput screens,
- High-content imaging (HCI) (i.e., evaluate more than 500 phenotypical parameters),
- Dose determination,
- Efficacy quantification,
- Combination strategies,
- PDX model selection, and
- Co-cultures for immuno-therapy applications.
Since PDXOs are biologically equivalent to PDXs, researchers can readily leverage large collections of well-characterized PDXOs (i.e., biobanks) as surrogates for heterogenous cancer patient populations in high throughput drug screens. This shrinks costs and timelines compared to in vivo studies, while maintaining clinical relevance. Compounds identified in PDXO panel screens allow researchers to make decisions earlier as opposed to waiting for late-stage in vivo PDX model studies, and to progress to more targeted in vivo efficacy studies with higher predictive confidence of the in vitro data.
Advantages and Challenges
As noted earlier, 3D in vitro tumor organoids are more clinically relevant than traditional 2D cell lines, while they can often save researchers time and money when compared to in vivo models. Moreover, since most available PDO/PDXO models are also available as PDXs, researchers have direct access to responsive in vivo matched models that can be used complementarily to allow for an efficient transition from early in vitro studies to late-phase animal-based validation trials.
The advantages of using PDOs is highlighted in a study that leveraged them to uncover an innovative therapeutic clinical candidate. In the study, Herpers et al. (supported by scientists at Crown Bioscience) used PDOs in combination with HCI to functionally evaluate complex drug responses. High throughput screening with tumor organoids identified a bispecific antibody that triggers EGFR degradation specifically in LGR5+ cancer stem cells, with minimal toxicity to healthy LGR5+ colon stem cells. The effect of the antibody was further validated in matched in vivo PDOX and PDX models. This example highlights the potential of functionally screening PDO panels with HCI to efficiently uncover drug candidates, while the subsequent in vivo studies in both PDOX and PDX models provides translational confidence in the PDO screening results.
Researchers should also be aware of some of the common challenges associated with tumor organoids. For instance, they can be developed only for solid tumors of epithelial origin, and they lack a tumor microenvironment component, albeit this limitation can be overcome with tumor organoid co-cultures. Co-cultures with non-autologous human immune cells are the most common and are being used to test the allogeneic T cell response/killing of tumor organoids and potency of immune checkpoint inhibitors.
Summary
Tumor organoids developed using HUB protocols faithfully recapitulate the complexities observed in the parental human cancer tissue, and they show high patient-relevance and translatability. The underlying nature of tumor organoids also makes them robust and reproducible across a wide variety of applications important for drug discovery, including high throughput assays, providing efficiency advantages over in vivo PDX models which are better suited for later stage validation studies.
Leveraging well-characterized tumor organoid biobanks and complementary PDX models, researchers are equipped with an in vitro system that bridges the gap between the convenience of traditional 2D cell lines and the predictive power of in vivo models. This can maximize the chances of selecting drug candidates with high translatability and clinical success.