 Explore the main applications for tumor organoids in preclinical cancer drug development, driven by the unique features of these models.
Explore the main applications for tumor organoids in preclinical cancer drug development, driven by the unique features of these models.
Tumor Organoids in Oncology Drug Development
Oncology agents continue to have the highest failure rates across all therapeutic areas. While several factors are responsible for driving this high failure rate, there is a confirmed need for more clinically relevant preclinical models. This is partially being fulfilled by tumor organoids within in vitro drug development. The rise of organoid technology is revolutionizing in vitro predictivity over existing 3D and 2D in vitro models, as well as guiding researchers to optimal in vivo models for late stage studies.
4 Applications of Tumor Organoids in Oncology Drug Development
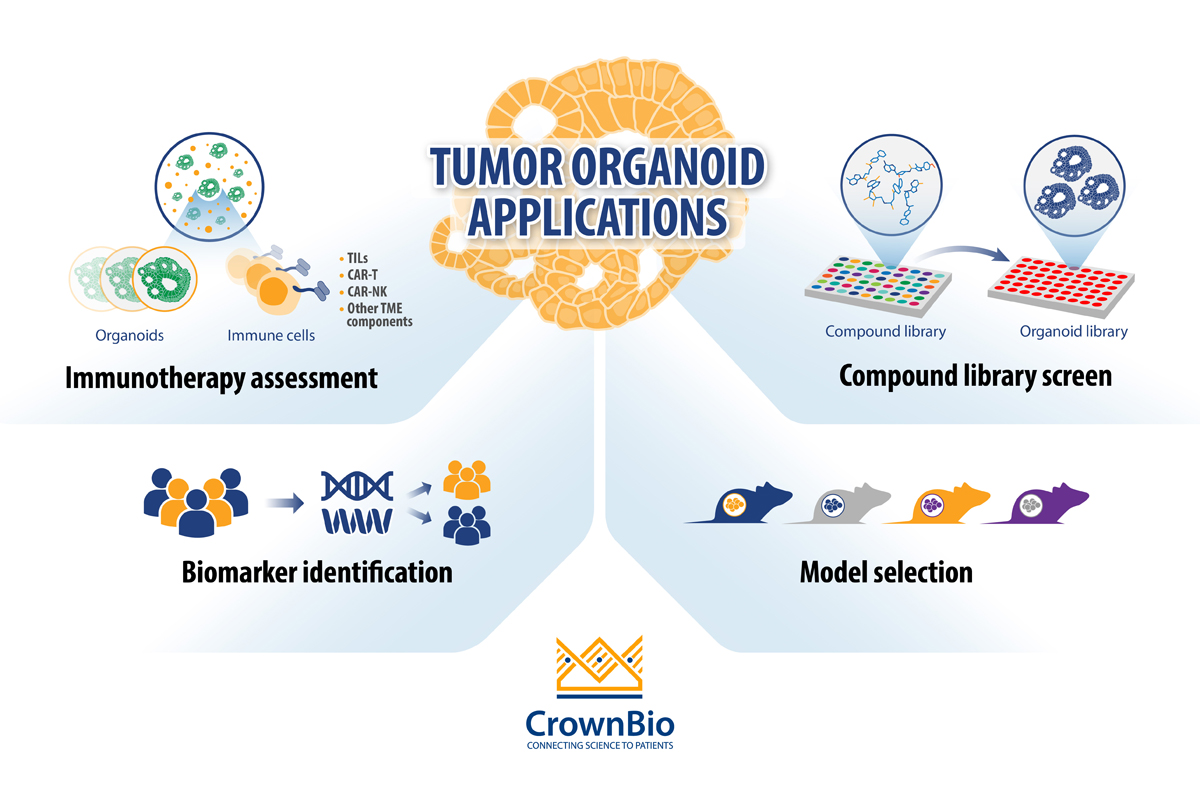
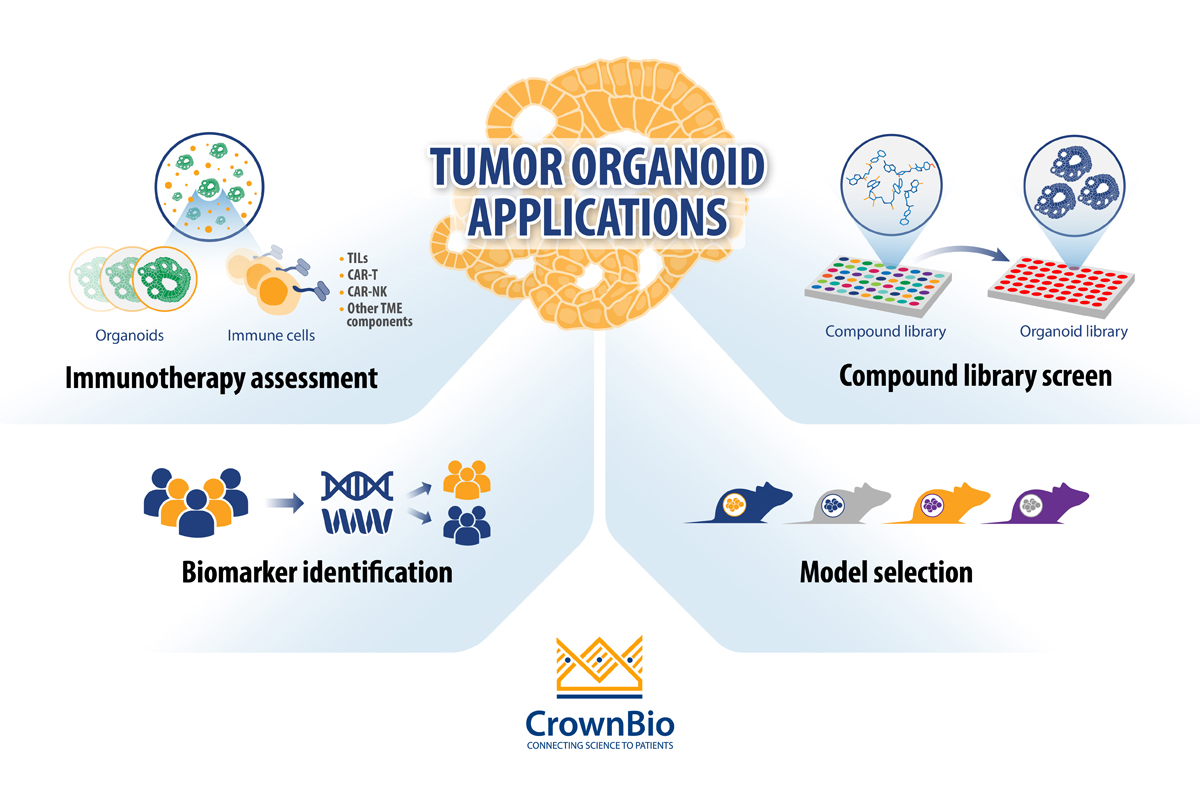
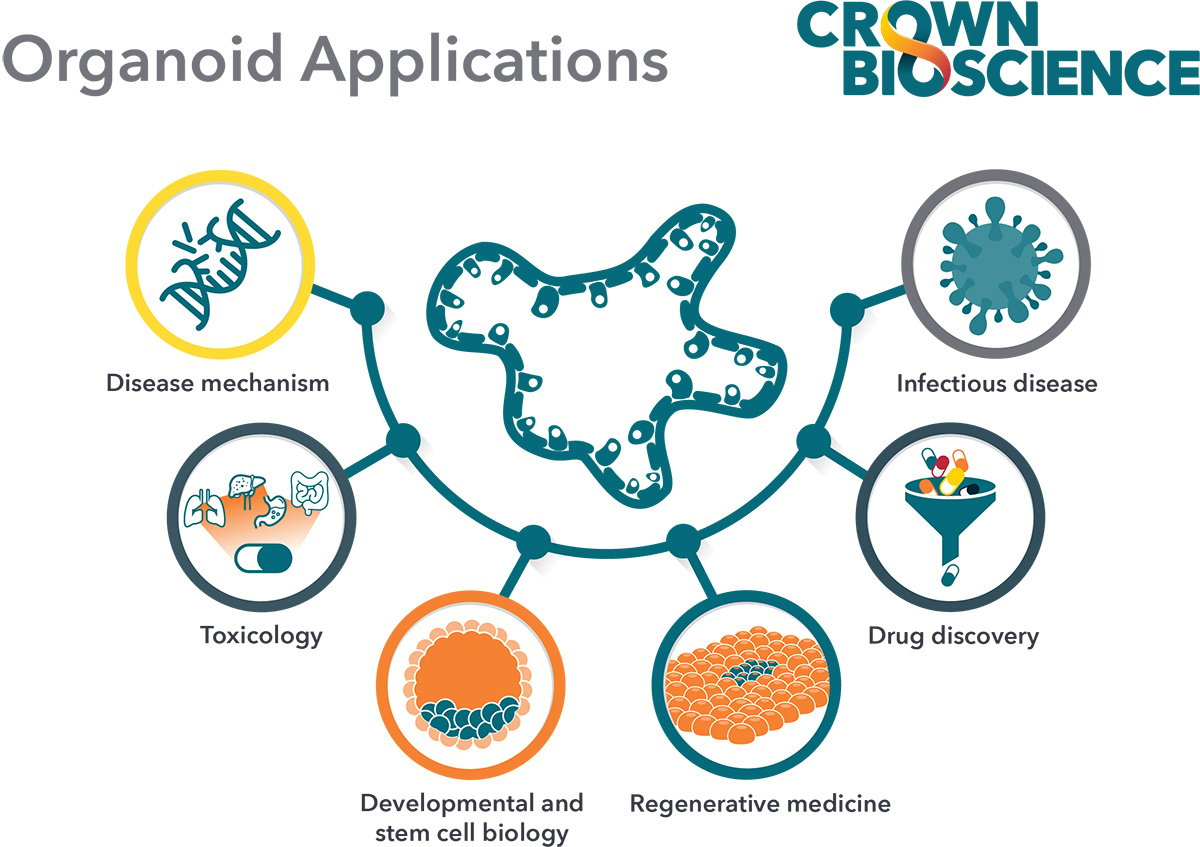
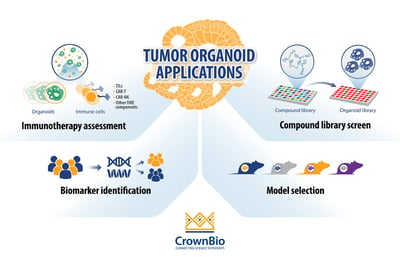
The current main applications for tumor organoids in cancer drug development are:
- Compound library screening: By using extensive and well-characterized tumor organoid biobanks, the patient relevance of in vitro “hits” is enhanced and quickly identifies lead agents that can be transitioned rapidly to relevant in vivo models.
- Biomarker identification: Differentially expressed biomarker candidates are evaluated by comparing the genomic mutational status of tumor organoids versus their matched healthy counterparts, or tumor organoid subtypes (derived from different cancer subtypes) with each other. By using biobanks of tumor organoids, the phenotypic and genetic diversity observed in patient populations is captured and drug response can be correlated to mutational status.
- In vivo model selection: By using collections of tumor organoids (biobanks), researchers can incorporate molecular and phenotypic profiles into their in vivo model selection process, increasing the applicability of the PDX model(s) selected.
- Immunotherapy assessment through co-culture with immune cells: Tumor organoid co-cultures are being developed for immuno-oncology applications to offer another preclinical platform that is more clinically relevant compared to cancer cell lines. Tumor organoid co-cultures have been developed using both autologous and non-autologous cells.
Main Features of Tumor Organoid Models
These expanded applications are being fueled by the main attributes of tumor organoids. All preclinical models have their own pros and cons, but there are some distinct features of tumor organoids that set them apart from other 3D culture systems, and make them the most clinically relevant in vitro option available today.
The main attributes that set organoids apart from other 3D in vitro models are:
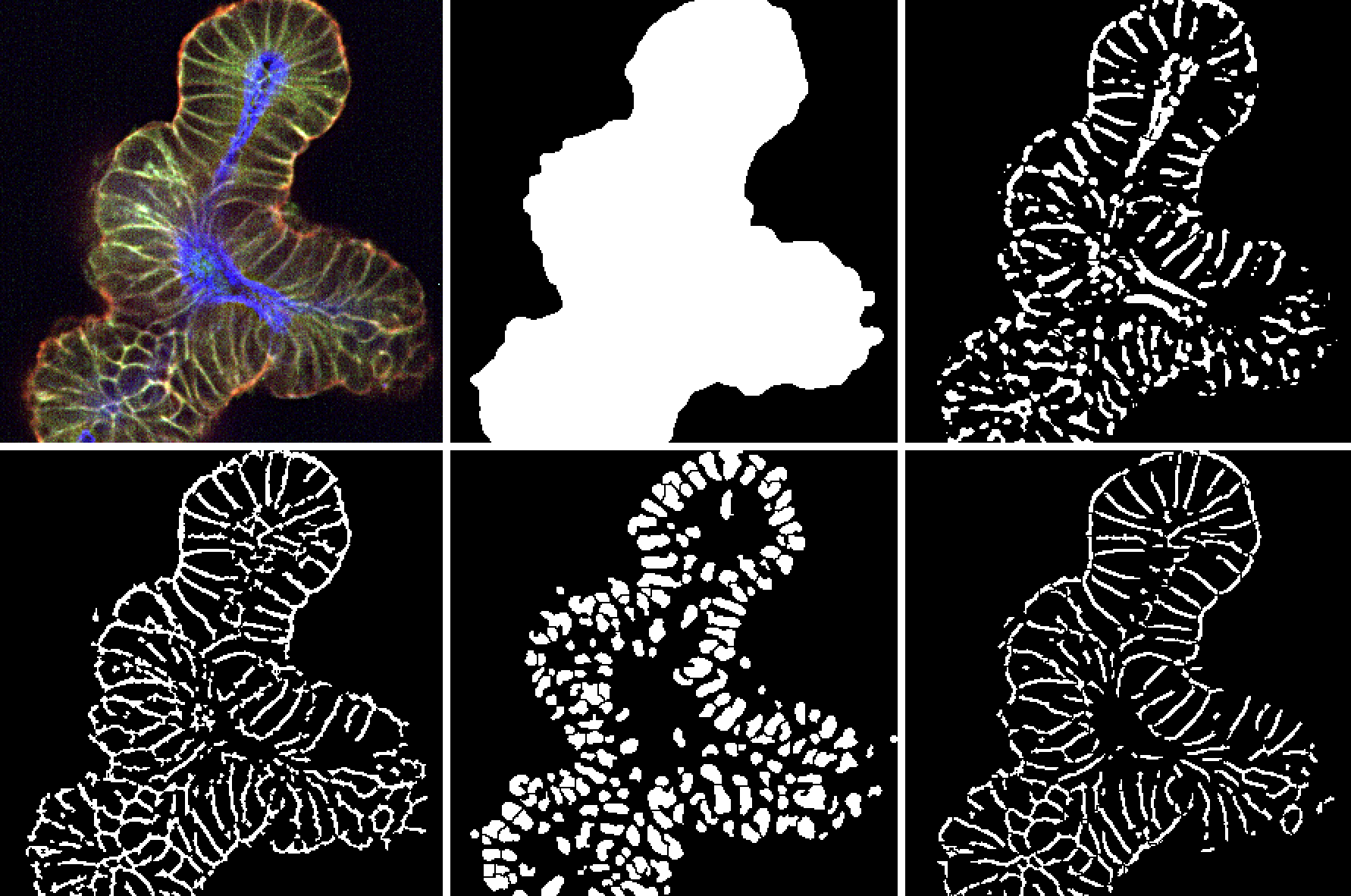
- Origin of the 3D structures: Organoids derive from stem cells present in adult epithelial tissues. Thanks to specific culturing conditions, these stem cells are able to self-renew and differentiate in vitro, giving rise to self-organizing multicellular 3D structures. Similarly to healthy organoids, tumor organoids faithfully recapitulate the complexities observed in human cancer tissue, including cancer stem cells (CSCs) and their downstream differentiated progeny.
- Nature of the driving force: Since organoids are derived from stem cells, their formation is driven largely by intrinsic developmental programs which leads to a remarkable recapitulation of tissue morphology and organ architecture.
- Length of time 3D cultures can be maintained: Organoids can be successfully cultured in vitro over long periods. Importantly, even after passaging, expansion, and cryopreservation, organoids retain the phenotypic and genetic features of the original organ or tumor over multiple generations.
- Recapitulation of the complexities of tumor tissue: Tumor organoids, established using exclusive HUB protocols, faithfully recapitulate key phenotypic and genetic features observed in the original tumor. HUB protocols also prevent the rapid exhaustion of CSCs which allows for their long term maintenance in vitro.
- Matched in vitro and in vivo cancer drug research platform: Biobanks of tumor organoids are being developed from patient biopsies or libraries of well-annotated PDXs. This provides a unique preclinical platform for cancer drug discovery and development. Researchers are using biobanks of tumor organoids for large-scale drug screens and testing drug combinations, as well as to allow easy transition to in vivo models for late stage investigations.
Conclusion
Tumor organoids are the most clinically relevant in vitro model for oncology studies based on unique model features. These features are enabling new applications for tumor organoids in oncology drug development, to enhance predictivity in early in vitro drug development phases and to facilitate transition to in vivo models for late stage studies.
Overall, within these applications, the higher patient relevance of tumor organoids is producing more accurate data on the efficacy and potency of investigational anticancer agents, ultimately improving the odds of success in downstream studies.