At ENA 2018 we’re presenting new data on our wide range of immuno-oncology (I/O) platforms, with four posters covering murine I/O models. This blog post reviews our posters on orthotopic syngeneic and prostate cancer tumor homograft models, as well as characterization and TIL analysis for both model types comparing subcutaneous and orthotopic implantation.
At ENA 2018 we’re presenting new data on our wide range of immuno-oncology (I/O) platforms, with four posters covering murine I/O models. This blog post reviews our posters on orthotopic syngeneic and prostate cancer tumor homograft models, as well as characterization and TIL analysis for both model types comparing subcutaneous and orthotopic implantation.
Prostate Homograft Tumor Models for Combinatory Immunotherapies
ENA Poster 250: Establishment of Mouse Prostate Homograft Tumour Models for Efficacy Evaluation of Combinatory Immunotherapies
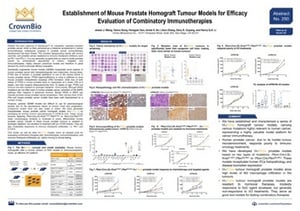 Our novel tumor homograft platform is featured in many of our ENA 2018 posters. Tumor homograft models combine the molecular and targeted benefits of GEMM with an improved operational simplicity for easier efficacy studies.
Our novel tumor homograft platform is featured in many of our ENA 2018 posters. Tumor homograft models combine the molecular and targeted benefits of GEMM with an improved operational simplicity for easier efficacy studies.
ENA Poster 250 focused on our new prostate cancer homograft models for immunotherapy and combination regimen development.
Preclinical Prostate Cancer Drug Development
Prostate cancer desperately needs new treatment options, and the most promising are combination regimens including immunotherapies (which, other than Sipuleucel-T, have not fared well in this cancer type), chemotherapies, and targeted agents.
Preclinical models are therefore needed which can test a variety of these combinations. They need to be immunocompetent models to allow I/O agent development, but also models which capture common mutations and molecular features of human disease development.
Genetically-engineered, immunocompetent prostate cancer models exist which replicate human disease features, but these aren’t great for efficacy studies as tumors develop at different times. Compound mutant mice have also been well-characterized but can be costly to breed.
Prostate Cancer Tumor Homograft Models
Our tumor homograft models are created by transplanting these well-characterized primary mouse tumors subcutaneously to syngeneic recipients. The resulting prostate cancer models, detailed in our poster, feature Pbsn-Cre;LSL-KrasG12D/+;Ptenflox/flox or Pbsn-Cre;Ptenflox/flox. The models are immunocompetent, morphologically similar to moderate-to-poorly differentiated human disease, and operationally easy to use for efficacy studies.
The poster features characterization and mutation load data (tumor homografts have a lower mutation load than syngeneics, closer to that of human disease), before moving on to efficacy studies with hormonal and targeted agents, and chemo- and immunotherapies.
Response across the three models and treatment modalities was mixed. The models were generally resistant to hormonal treatments, with a better response to an mTOR inhibitor. The response was poor to common immune checkpoint inhibitors (anti-PD-1 and CTLA-4), confirming that combination regimens may be the way forward for immunotherapy in prostate cancer
In summary, tumor homograft models provide a novel platform for prostate cancer immunotherapy drug development, providing clinically relevant features and immunocompetence in an easy to use model.
Syngeneic Models in I/O Drug Discovery
ENA Poster 241: Utilising Murine Syngeneic Model Systems in I/O Drug Discovery: Broad Applications and Guidance on Model Selection and Data Interpretation
 Our next poster features syngeneic models. We’ve often presented on MuScreen™ – the first and most experienced in vivo syngeneic screening platform for immunotherapy development. While previous posters looked at efficacy results, here we focused on in-depth syngeneic model characterization to help with study design and model selection.
Our next poster features syngeneic models. We’ve often presented on MuScreen™ – the first and most experienced in vivo syngeneic screening platform for immunotherapy development. While previous posters looked at efficacy results, here we focused on in-depth syngeneic model characterization to help with study design and model selection.
Syngeneic Model Characterization
Detailed syngeneic model characterization is needed to truly understand the interplay between immunomodulation and antitumor phenotype. This characterization needs to capture the dynamic changes in immune cell presence and infiltration across the different compartments of tumor-bearing mice, all after treatment with checkpoint inhibitors.
To characterize our MuScreen syngeneic models, we treated them with anti-PD-1 antibody and then evaluated antitumor efficacy and the immunophenotypic changes in blood, tumor, and other immune organs. Our ENA 2018 poster features initial data on basal levels of immune cell subpopulation tumor infiltration and immunoprofiling data from selected tumor models.
Syngeneic Immunoprofiling Results
Our comprehensive syngeneic model characterization revealed some interesting results. Variation was seen for many features – for basal level immune cell infiltration for each model, and for individual mice immunophenotypic changes in response to PD-1 blockade.
Each of the syngeneic models also have variable levels of immune cell infiltration. These data could guide researchers in choosing models for studies, based on the mechanism of action or proposed target immune cells of a given I/O agent.
For models with robust tumor growth inhibition after anti-PD-1 treatment, there was a trend towards meaningful immunomodulatory changes. However, we didn’t see this for models which were less responsive to treatment, or treatment-resistant.
Overall, the characterization data we are starting to compile is revealing dynamic changes in immunoprofiling in relation to antitumor response. These data could help make strategic decisions on syngeneic model selection as well as which drugs to carry forward for combination testing.
Recapitulating the Tumor Microenvironment with Orthotopic Syngeneics
ENA Poster 329: Recapitulating the Orthotopic Tumour Microenvironment using Bioluminescent Syngeneic Models in Immune Competent Mice for Checkpoint Inhibitor Interrogation
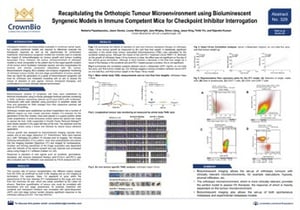 Another way we’re making syngeneic models more relevant in drug development is with orthotopic models. Response to immunotherapy can be heavily dependent on the tumor microenvironment (TME), and orthotopic syngeneics have a more patient-comparable TME due to their tumors’ organ-specific location. This allows metastatic spread and better recapitulates the immune and stromal components of the TME. Overall, this can provide a more clinically-relevant model for immunotherapy assessment.
Another way we’re making syngeneic models more relevant in drug development is with orthotopic models. Response to immunotherapy can be heavily dependent on the tumor microenvironment (TME), and orthotopic syngeneics have a more patient-comparable TME due to their tumors’ organ-specific location. This allows metastatic spread and better recapitulates the immune and stromal components of the TME. Overall, this can provide a more clinically-relevant model for immunotherapy assessment.
Bioluminescent Syngeneic Models
We’ve developed the orthotopic syngeneics detailed in this poster as bioluminescent models. This allows non-invasive longitudinal monitoring of tumor burden, and end stage quantification of tumor spread.
The poster mainly focuses on the Hepa 1-6-lux-model, detailing treatment with standard of care sorafenib and an anti-PD-1 antibody. Treatment with sorafenib significantly reduces whole body tumor associated bioluminescence, and a trend in reduction is also seen for PD-1.
Metastasis Models
Another benefit of bioluminescent imaging is the easy quantification of metastasis. The poster also features the 4T1 model, which metastasizes to the lungs and bone following implantation in the mammary fat pad.
In summary, using orthotopic syngeneic models provides a more clinically relevant TME for immunotherapy testing and allows the easy set-up of both spontaneous metastasis and experimental metastasis models.
Infiltrating Immune Cell Patterns Differ in Orthotopic Homograft Tumors
ENA Poster 313: Orthotopic Homograft Tumours (Syngeneic) Display Distinct Tumour Infiltrate Immune Cell Patterns from that of Subcutaneous Counterparts
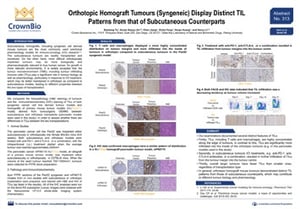 Our final poster on murine immuno-oncology models stays with orthotopic syngeneic models, looping back to immunoprofiling as well as tumor homografts.
Our final poster on murine immuno-oncology models stays with orthotopic syngeneic models, looping back to immunoprofiling as well as tumor homografts.
As discussed above, subcutaneous models are commonly used for preclinical I/O pharmacology studies, but orthotopic tumors can be more human disease-relevant while maintaining a better tumor microenvironment, including tumor infiltrating immune cells.
This poster compares TILs for pancreatic syngeneic and tumor homograft models, implanted either orthotopically or subcutaneously.
Distinct TIL Patterns of Orthotopic Tumors
Histopathology and IHC results show a definite difference between TIL patterns for orthotopic and subcutaneous sygeneic and tumor homograft models. The main difference is that TILs (e.g. T cells and macrophages) are significantly more infiltrated into the inside of orthotopic tumors compared with subcutaneous.
Interestingly, for subcutaneous tumors, treatment with checkpoint inhibitors such as anti-PD-1 or anti-CTLA-4 causes an increase in TIL infiltration from the tumor margin to center.
We also saw fewer TILs in large tumors compared to smaller ones, regardless of orthotopic vs subcutaneous implantation.
Overall, distinct TIL patterns for orthotopic models may contribute to different tumor biology and pharmacology, which should be considered in immunotherapy assessment studies.
More on ENA 2018
Don’t miss our 10 posters at ENA this week, or our posts on new humanized models and PDX presentations at ENA 2018.