 Learn more about patient-derived organoids, a unique ex vivo tool enabling personalized cancer research for improved treatment of metastatic disease.
Learn more about patient-derived organoids, a unique ex vivo tool enabling personalized cancer research for improved treatment of metastatic disease.
Challenges of Tumor Heterogeneity and Metastases
Although cancer research and treatments have advanced over the past decades, many cancers remain difficult to treat or incurable. This is due to a variety of factors, including lack of effective therapies and tumor heterogeneity.
Several published studies support the concept that tumors show exceptional heterogeneity between patients, between tumors from the same patient, and even within a single tumor. Tumor heterogeneity is caused by genetics, epigenetic factors, or both combined.
A further issue is metastasis. Cancers are not a single disease, they are highly dynamic and evolve resistance over time, including to advanced therapeutics. These characteristics make cancer nearly impossible to cure once it spreads to other organs.
Once metastases are diagnosed, the major goal of cancer therapy is disease maintenance, and keeping tumor burden at a minimum to increase survival time. More than 90% of cancer deaths are attributed to metastasis and drug resistance.
These factors are driving a need for personalized medicine, with more individualized research and treatment regimens. Novel preclinical models are needed to recapitulate all the above disease features, both for conventional drug development and a more advanced personalized approach.
Current Cancer Models
A long-standing challenge in cancer research is the development and application of experimental human cancer models that accurately reflect tumor heterogeneity, diversity, disease progression, metastasis, therapeutic response, and drug resistance.
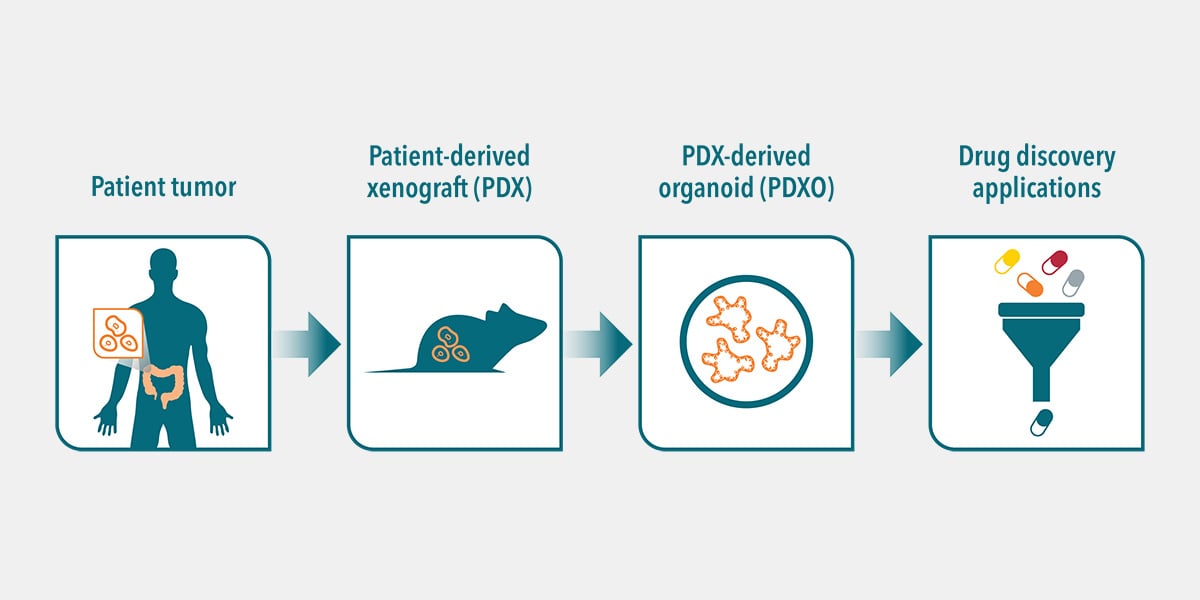
There are many different preclinical cancer models available, all with different strengths and optimal uses, including patient-derived xenografts (PDX). PDX more accurately represent human tumor heterogeneity, genomics, metastatic behavior, and concordance of drug response with patient tumor response.
PDX models are ideal for more predictive, preclinical studies, simulating Phase II-like mouse clinical trials and representing patient diversity. However, they’re not readily amenable for high throughput screening of large numbers of agents. PDX model establishment, as well as low establishment rates for some cancer types, also present some obstacles for a personalized approach.
New advancements in 3D ex vivo culture technologies have now created new drug development tools, such as organoids, specifically to simulate the clinical drug response in the lab. Development of these advanced and sophisticated 3D ex vivo models is critical and important for efficient, fast translation of preclinical research into clinical treatment regimens.
Patient-Derived Organoids in Personalized Cancer Research
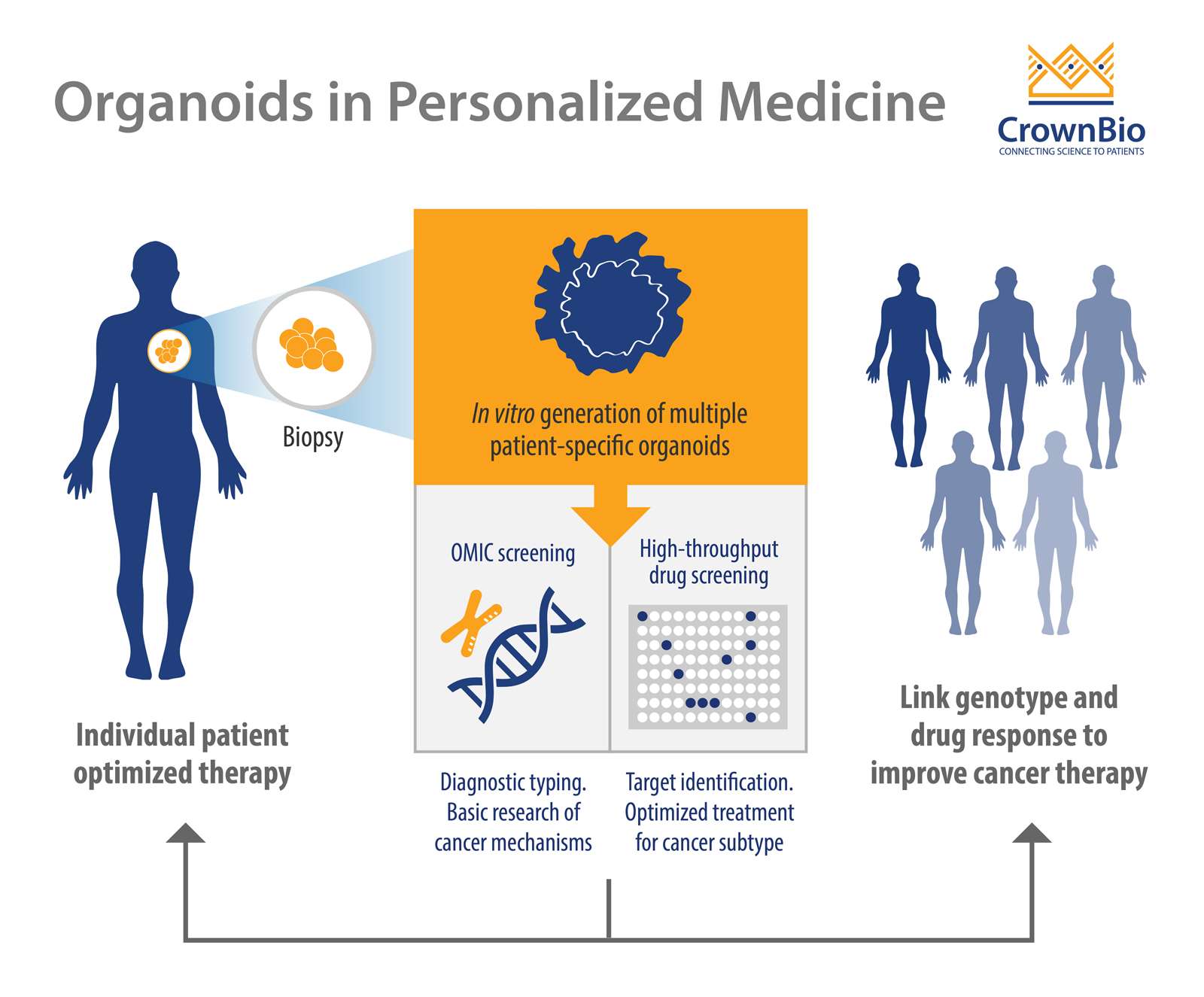
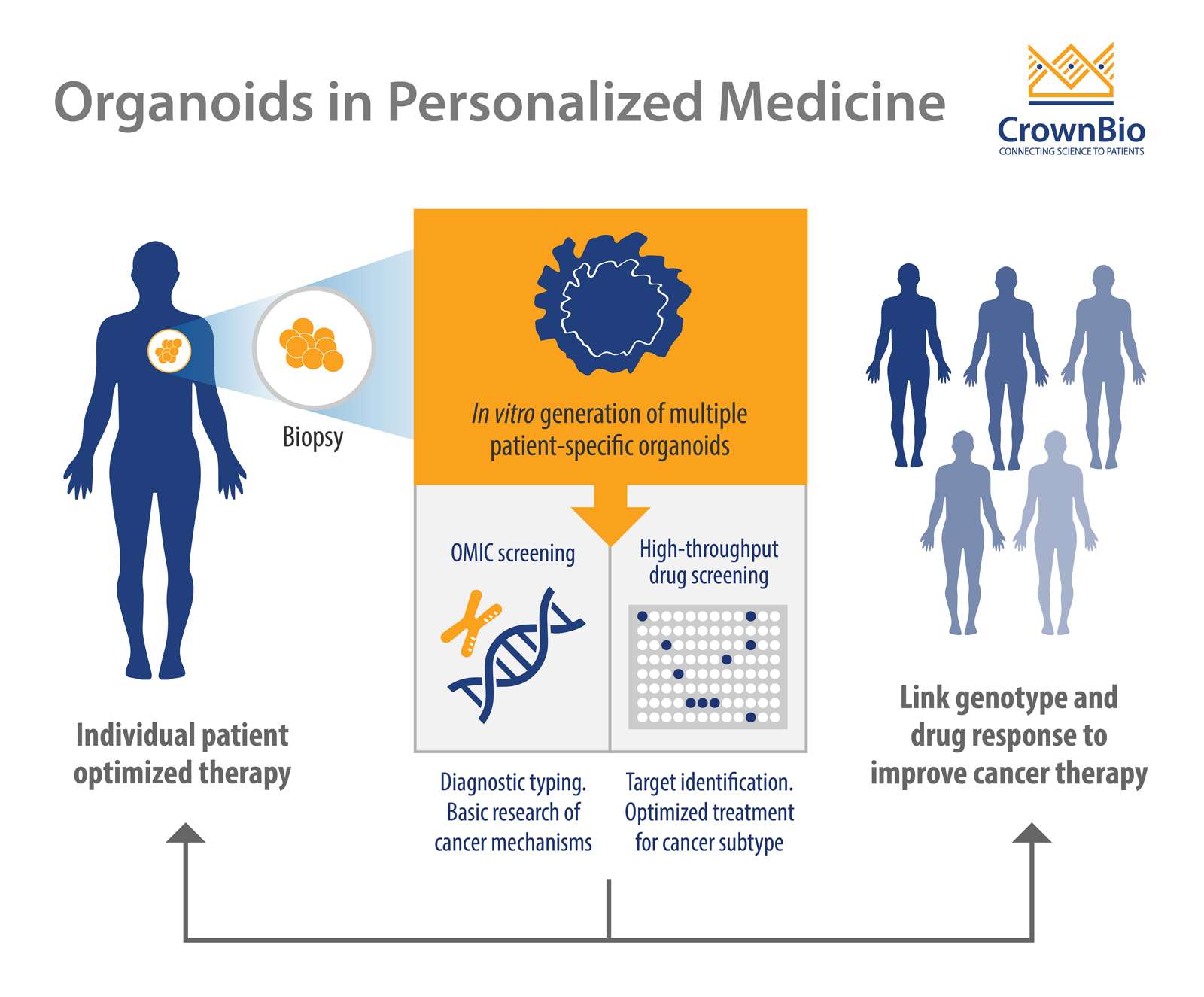
PDOs are advanced 3D, ex vivo cell cultures from patient tissues, containing stems cells to enable self-renewal, organization, differentiation, and disease progression. They mimic the original tumor in multiple ways, including genomically and histologically, providing an ideal tool for personalized cancer therapy development, and metastatic disease research.
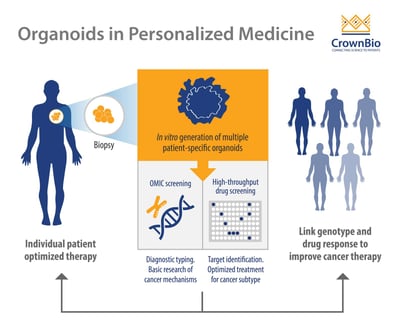
Personalized therapy is an advanced and novel strategy which identifies the right treatment for the right patient by determining the patient’s tumor genetic signature. PDOs have several advantages compared to pre-existing models, which make them an invaluable tool in personalized cancer therapy.
Ex vivo PDOs can be quickly generated with high efficiency from both patient-derived healthy and tumor tissues (including metastatic disease) and validated with histology, genomics, and drug response. This is in contrast to current in vivo models which are more expensive, time consuming, and labor intensive to develop.
PDOs can be expanded ex vivo enabling high throughput drug screening, patient-specific drug testing, and the development of personalized treatment.
A final major advantage is that organoids can be grown from limited amount of starting materials (biopsies) and used for drug screening purposes.
While organoids do have many advantages, there are still some limitations to consider, including a lack of immune system in the organoid cultures which can limit immuno-oncology use. Some organoids cannot be expanded for long time periods, and growth factors/inhibitors in the culture medium may affect the gene expression pattern in organoids.
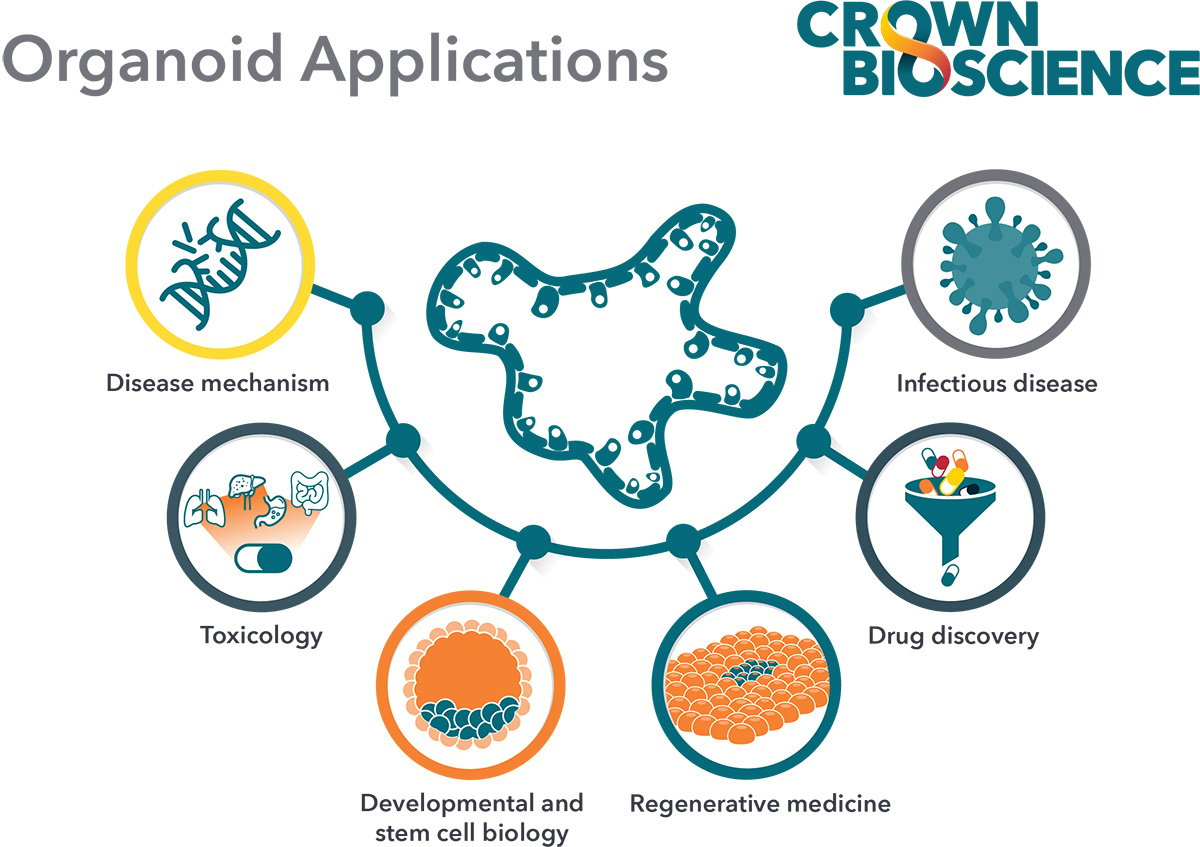
Organoids in Preclinical Research
Several researchers have proved the value of these “tumor-in-a-dish” approaches in personalized cancer therapy, both for preclinical drug screening and prediction of patient treatment outcome. Studies include organoids grown from metastatic tumors, from patients with gastroesophageal or colorectal cancers. The patients were enrolled in a variety of clinical trials; with the experimental regimens they were given also tested on their organoids. The results showed a clear correlation between organoid and patient response.
Recently, Alice Soragni’s group developed a method of mini-ring organoid screening technology which allows the development of organoids and high throughput screening within a week after patient surgery. This time line is realistically compatible for therapeutic decision making in the personalized treatment of cancer patients.
Summary
PDOs are an attractive ex vivo model system for studying tumor evolution and response to drugs and treatments. They provide a novel tool for personalized cancer research, where lab-grown mini tumors offer a way to test many drugs in parallel. For individual treatments, this saves time and allows the testing of multiple drugs which is not possible within recurrent or metastatic cancer patients, potentially extending lifetimes.
In the future, organoids could help to transform many aspects of medical research from basic disease understanding through to drug development and personalized treatment for the patients.